In vivo gene therapies have delivered astonishing results where few or no treatments have existed previously. Despite their curative potential, the field of gene therapy has not seen effective commercialization. Here, we compare adeno-associated virus (AAV) viral vectors and lipid nanoparticle (LNP) nonviral vectors at different phases of the product life cycle to understand why.
While LNPs may face greater preclinical development hurdles than AAV, they are expected to have more streamlined clinical development and commercialization pathways, which will allow them to extend the scope of clinical indications accessible for treatment by in vivo gene therapies.
The emergence of nonviral vectors represents an opportunity to propel in vivo gene therapy further into commercial adoption and expand its impact globally. LNPs are poised to become the first commercially approved nonviral vectors for in vivo transgene delivery and genome editing approaches.
Preclinical development
LNPs are attractive for their versatile and unrestricted cargo capacity. Whereas AAV can deliver DNA just under 5 kb, LNPs can encapsulate a variety of cargo including mRNA, DNA, siRNA, and proteins, even in combination, with as yet unrestricted cargo capacity. Additionally, LNPs’ transient expression profile, which for some transgene delivery applications may be less desirable, is considered a boon for genome editing from a safety perspective. For this reason, LNPs are potentially more well suited for genome editing applications which have larger cargo to achieve targeted, safe, and precise editing of the genome.
However, initial formulation development of LNP vectors is frequently challenged by a lack of model and structure-function relationship. To add to this challenge, mRNA-LNP formulation development is cargo- and target-dependent, necessitating screening of different lipid and mRNA ratios; even an off-the-shelf LNP formulation will require optimization. Often, high throughput approaches are leveraged utilizing microfluidics, robotics, or barcoding to increase the speed with which candidate formulations can be identified. Then, several formulations may still need to be screened across nonclinical disease-relevant models to assess biodistribution and safety. If a novel lipid is leveraged, additional safety testing will be required to characterize the safety profile of the novel excipient. To add to this, biodistribution in some nonclinical models is not predictive of biodistribution in humans.
The LNP supply chain is also complex. GMP lipid synthesis can require different synthesis pathways due to availability of precursor reagents, high solvent levels, heat, and process residuals. Furthermore, purification can be challenging and biodegradability can impact manufacturing yields. On the bright side, LNP manufacturing is straightforward and much faster compared to other CGT manufacturing process. The amount of process variability and controls required for AAV production are significantly more extensive, driven by biological process residuals and safety concerns, resulting in more process challenges and higher COGS.
Clinical development
From a clinical point of view, safety and targeting are the hot topics. Arguably the most critical challenge limiting clinical adoption of LNPs for in vivo gene therapy is improved biodistribution via systemic delivery. LNPs do not possess innate tropism, are subject to non-specific serum protein interactions, and are consequently susceptible to uptake by the reticuloendothelial system. These factors all amount to preferential accumulation to the liver, and clinical in vivo gene therapy LNP applications thus far have been limited to targeting the liver, lungs, or blood. However, in vivo nonviral gene therapies in preclinical development expand that list to include ocular, lung, CNS, solid tumor and cardiovascular targets. This is often accomplished by conjugating the LNPs with antibodies, peptides, and other ligands that target tissue-specific antigens, as well as innovating upon novel lipid chemistry. Still, significant development is needed to achieve in vivo targeted delivery to other organs, necessitating innovation in LNP design to enhance trafficking across biological barriers.
However, LNPs possess an excellent safety profile, especially in relation to their viral counterparts. No need to assess potential for persistent expression or insertional mutagenesis, no concerns with pre-existing neutralizing antibodies, and the ability to re-dose LNPs all contribute to their appeal from a clinical safety perspective, as well as having a favorable impact on development costs and complexity.
Commercial phase
LNPs edge out AAV from a cold chain storage and distribution angle. The majority of AAV drug products must be stored at -60° C, whereas LNPs have been successfully lyophilized which can significantly reduce cold chain supply burden. The impact of storage at higher temperatures is increased access to patients beyond major metropolitan medical centers in western nations, which could allow for true globalized patient access and improved patient equity. Not only would this increase the clinical impact of in vivo gene therapies, but it would also result in a larger addressable patient population which allows developers to achieve more favorable ROI and manufacturing economies of scale.
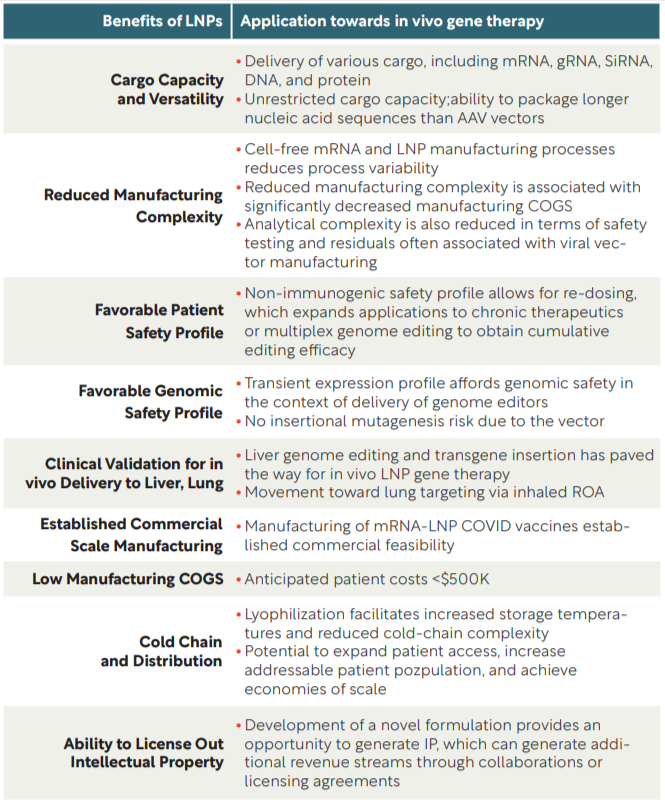
Table 1
Benefits of LNPs for in vivo gene therapy applications
And though the LNP intellectual property landscape is complex and presents a potential barrier to entry — with mRNA, lipids, and the LNP formulations being readily subject to patents — the flip side is that development of a novel formulation provides an opportunity for developers to generate novel IP, with which they may pursue collaborations or licensing agreements to generate additional revenue streams.
From a regulatory perspective, there is some uncertainty around the designation of the lipids as an active ingredient, excipient, or drug substance in the LNP DP. While some may view lipids as inactive ingredients that stabilize the cargo, and for which a novel excipient classification would be appropriate, others view the lipids as essential to the function of the DP, justifying their classification as a drug substance. The regulatory expectations moving forward should be monitored, as they may impact the degree of control and testing required for these non-commodified reagents.
Widespread adoption
AAV represents the gold standard of in vivo gene therapy vectors, but LNPs are poised to become the first commercially approved nonviral in vivo gene therapy vector. Widespread adoption and commercial success of in vivo gene therapies will be contingent on the ability to bring to the market safe, cost-effective, and efficacious drugs. Despite its current challenges, LNPs are poised to meet this requirement more effectively than AAV.
Read the full whitepaper here.
Subscribe to our e-Newsletters
Stay up to date with news, articles and insights relevant to cell and gene therapy development and manufacturing. Plus, get special offers from Cell & Gene Therapy Review delivered right to your inbox!
Sign up now!