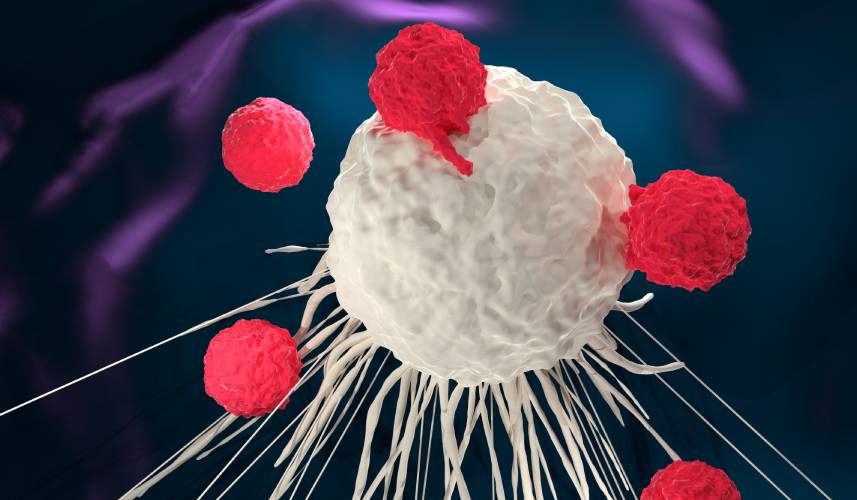
CAR-T cell therapy has shown promising results in treating blood cancers, such as multiple myeloma and certain forms of leukemia and lymphoma. The formulation of these immunotherapies involves firstly extracting T cells from a patient’s blood, and genetically modifying them in a laboratory to express a chimeric antigen receptor (CAR). These modified T cells are then infused back into the patient, where they target the cancer cells expressing the specific protein recognized by the CAR. This approach to treatment aims to help reduce cytotoxicity to non-target cells, leading to more effective treatment and fewer side effects.
Addressing roadblocks in CAR-T cell therapy manufacture
The current production of CAR-T cell therapies is predominantly centralized, requiring healthcare facilities to send patient samples to specialized manufacturers. This brings with it several significant challenges, for instance, the transportation of cells and final products between healthcare providers and manufacturing sites requires cold chain storage, resulting in substantial logistical expenses. In addition, the time between the initial cell collection and the return of the finished drug product to the hospital can extend to several months, delaying the start of urgent treatment.
Decentralizing the production of autologous CAR-T cells and taking it closer to the patient—such as within hospital laboratories—could mitigate these issues by eliminating long-distance transportation and its associated costs. This approach would also significantly reduce the turnaround time from cell harvesting to therapy administration, potentially to as little as three days, enabling earlier and more effective treatment. There is consequently a growing trend in North America and Europe for larger medical institutions to produce their own cell therapy products on site rather than outsourcing development. Regulatory bodies are increasingly acknowledging the unique nature of cell therapies compared to traditional pharmaceuticals, and are supporting the transition to decentralized manufacturing by offering exemptions to healthcare institutions that meet stringent GMP and quality control standards.
Click here to read the full article.
This article was originally published on Labcompare.
Subscribe to our e-Newsletters
Stay up to date with news, articles and insights relevant to cell and gene therapy development and manufacturing. Plus, get special offers from Cell & Gene Therapy Review delivered right to your inbox!
Sign up now!